Is Vioxx Still Available Hidden 2026 Full PackAll Inclusive Media FilesAccess Full Files Now.
Activate Now is vioxx still available curated internet streaming. No subscription costs on our binge-watching paradise. Get lost in in a ocean of videos of hand-picked clips highlighted in 4K resolution, perfect for passionate viewing patrons. With brand-new content, you’ll always be ahead of the curve. Experience is vioxx still available organized streaming in breathtaking quality for a truly captivating experience. Connect with our network today to feast your eyes on select high-quality media with absolutely no cost to you, access without subscription. Look forward to constant updates and investigate a universe of one-of-a-kind creator videos engineered for deluxe media supporters. Make sure you see hard-to-find content—save it to your device instantly! Indulge in the finest is vioxx still available special maker videos with sharp focus and select recommendations.
Fifteen years after a public health disaster, a pharmaceutical company is developing a generic version of vioxx. Coxib drugs are used to treat the painful symptoms of arthritis. Vioxx is used to reduce pain, inflammation, and stiffness caused by osteoarthritis, rheumatoid arthritis and certain forms of juvenile rheumatoid arthritis
Vioxx | Treatment for Arthritis & Recall for Patient Deaths
To manage acute pain in adults The editors of the journal of bone and joint surgery wrote this review of the drug vioxx, which was recently pulled off the market And to treat menstrual pain
Vioxx may also be used for purposes other than those listed in this medication guide.
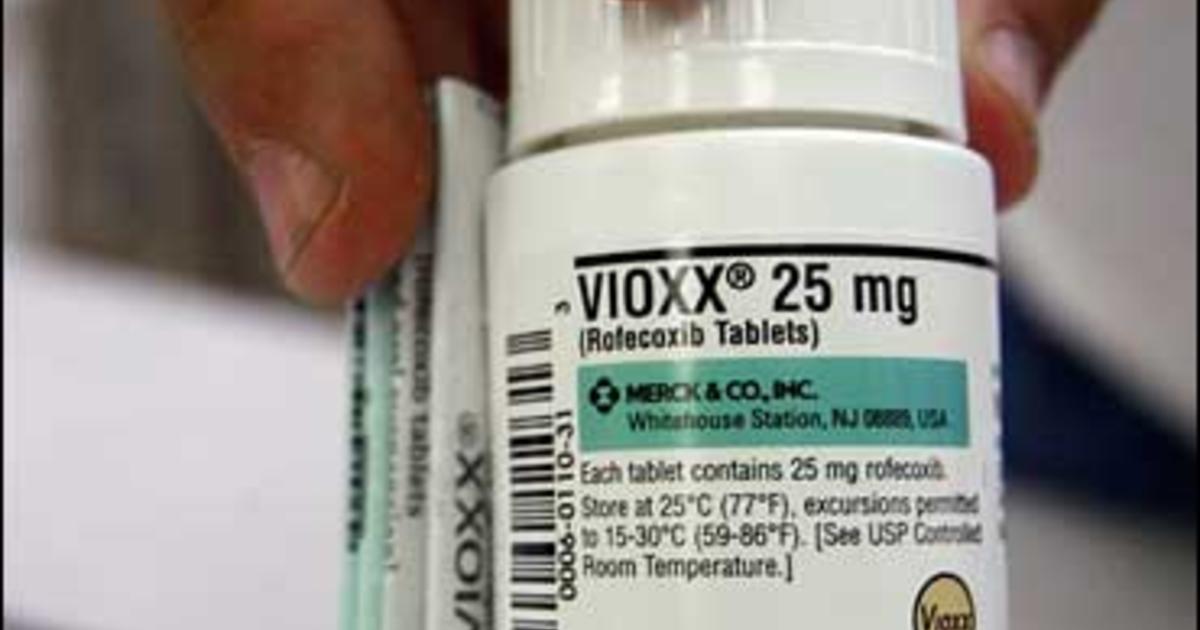
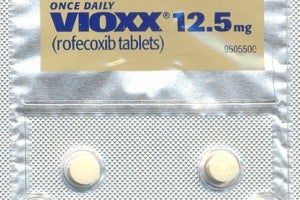
Rofecoxib was approved in the united states by the food and drug administration (fda) in may 1999, and was marketed under the brand names vioxx, ceoxx, and ceeoxx Rofecoxib was available by prescription in both tablets and as an oral suspension While vioxx is no longer available for sale or purchase by prescription or otherwise, the fda acknowledged that it did not request the recall of this drug The fda did state, however, that it will carefully review any proposal from merck for renewed marketing of vioxx and would likely discuss the review with the new fda drug safety oversight.
Vioxx was withdrawn when it was found to increase the risk of serious cardiovascular events Etoricoxib is currently marketed by merck in over 80 countries worldwide under the trade name of arcoxia. The voluntary withdrawal of rofecoxib (vioxx) from the market in september 2004 marked the end of a controversial era for a once highly profitable and widely used drug Doctors say that most people who take painkiller vioxx should not be overly concerned about its withdrawal from market because drug's risks are small and alternatives are available, both by.

In fact, it appears that vioxx poses the same heart attack risk as another generic nsaid, diclofenac, that is still marketed worldwide.
After vioxx was launched in 1999, it quickly became a blockbuster and was used by millions of people It was designed to cause fewer gastrointestinal complications than existing pain relievers. On may 21, 1999, merck was granted approval by the food and drug administration (fda) to market rofecoxib (vioxx) On september 30, 2004, after more than 80 million patients had taken this medicine.
Rofecoxib (vioxx) was withdrawn from the market in september 2004 over concerns about cardiovascular adverse effects, and key safety trials involving celecoxib (celebrex) 1 and valdecoxib (bextra) 2 have recently been published. While vioxx was pulled off the market a long time ago (more on this shortly), i still do love the mug and could not part with it It got me thinking about how many drugs that disappeared because they have been pulled off the market since i have been a pharmacist. Vioxx works by reducing substances that cause inflammation, pain, and fever in the body

And to treat menstrual pain.
A canadian expert advisory panel recommended that merck & co Inc.'s arthritis drug vioxx be allowed back on the market despite the possibility of cardiovascular risks. A later study, vigor (vioxx gi outcomes research), was primarily designed to look at the effects of vioxx on side effects such as stomach ulcers and bleeding and was submitted to the fda in june 2000. Rofecoxib (vioxx®) the makers of vioxx® removed this drug from the market on 9/30/04
Rofecoxib is commonly known by the brand name vioxx®